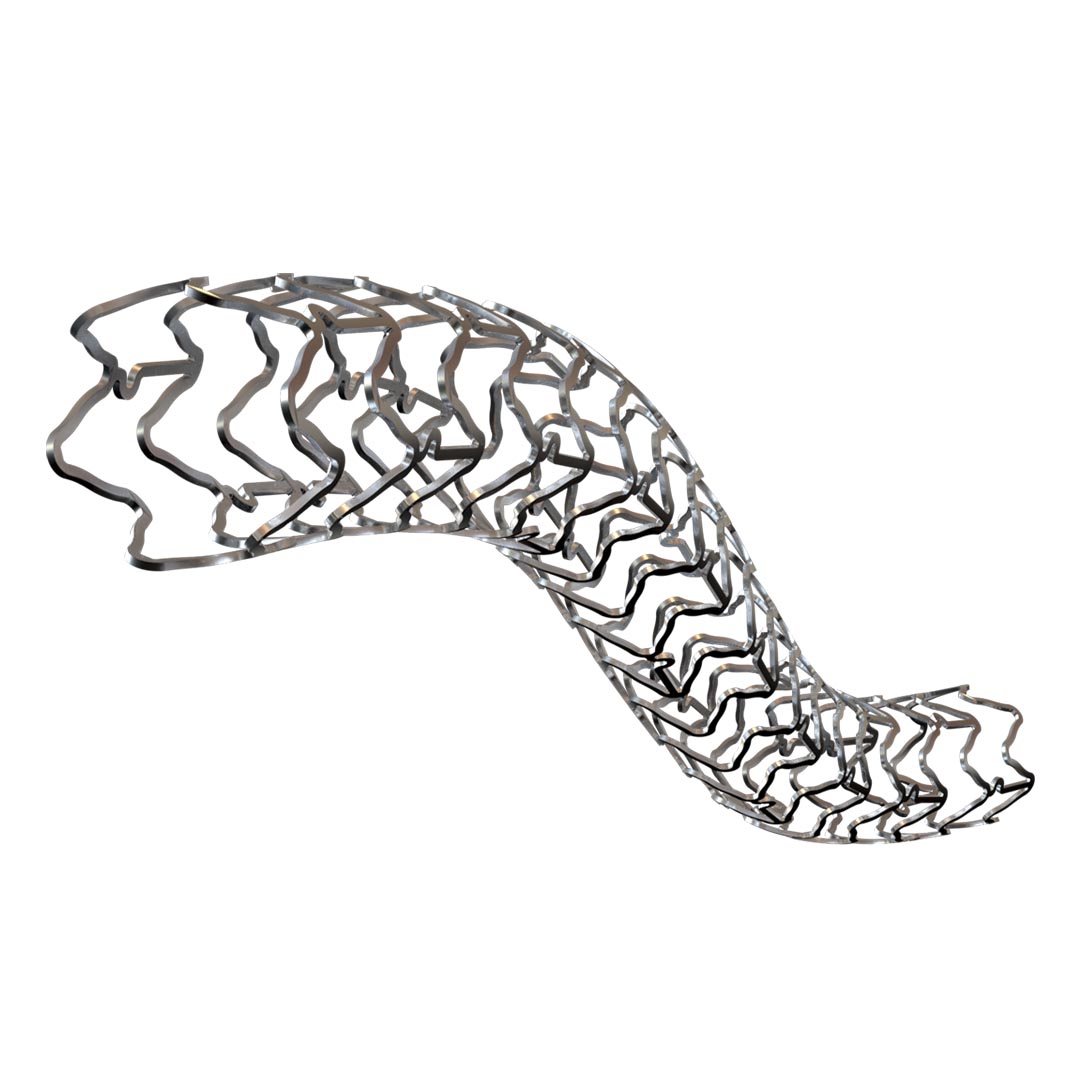
The ATLAS Drug Eluting Coronary Stent System SS is a stainless steel (SS) stent platform coated with a sirolimus formulation (1 µg/mm²), designed to prevent restenosis and support durable revascularization in patients with coronary artery disease. By delivering a targeted antiproliferative effect at the lesion site, it is indicated for both de novo coronary lesions and in-stent restenosis (ISR) management, thereby extending its clinical versatility in the percutaneous coronary intervention (PCI) setting.
- High Strength-to-Weight Ratio: Enables thinner struts without sacrificing radial force, ensuring ease of deliverability and vessel conformability.
- Proven Biocompatibility: Stainless steel has a long history of safe use in coronary interventions, supporting stable endothelization.
- Optimized Strut Geometry: Precisely formed lattice structure provides consistent drug distribution and minimal tissue trauma.
- Improved Crossability: The low-profile SS stent tracks smoothly through complex or calcified lesions, reducing the need for extensive lesion preparation.
- Targeted Antiproliferation: Gradual drug release inhibits smooth muscle cell proliferation, a primary contributor to in-stent restenosis.
- Enhanced Endothelialization: Balanced drug elution fosters early endothelial coverage, mitigating late stent thrombosis risk.
- Fluoroscopic Precision: Platinum-iridium markers at the stent’s extremities promote accurate stent positioning in the coronary vasculature.
- Reduced Geographic Miss: Clear visibility assists operators in precise stent deployment and lesion coverage.
Nominal pressure of 10 atm and a rated burst pressure of 20 atm provide interventional cardiologists the latitude to optimize stent expansion and device apposition based on lesion morphology.
- High Strength-to-Weight Ratio: Enables thinner struts without sacrificing radial force, ensuring ease of deliverability and vessel conformability.
- Proven Biocompatibility: Stainless steel has a long history of safe use in coronary interventions, supporting stable endothelization.
- Optimized Strut Geometry: Precisely formed lattice structure provides consistent drug distribution and minimal tissue trauma.
- Improved Crossability: The low-profile SS stent tracks smoothly through complex or calcified lesions, reducing the need for extensive lesion preparation.
- Targeted Antiproliferation: Gradual drug release inhibits smooth muscle cell proliferation, a primary contributor to in-stent restenosis.
- Enhanced Endothelialization: Balanced drug elution fosters early endothelial coverage, mitigating late stent thrombosis risk.
- Fluoroscopic Precision: Platinum-iridium markers at the stent’s extremities promote accurate stent positioning in the coronary vasculature.
- Reduced Geographic Miss: Clear visibility assists operators in precise stent deployment and lesion coverage.
Nominal pressure of 10 atm and a rated burst pressure of 20 atm provide interventional cardiologists the latitude to optimize stent expansion and device apposition based on lesion morphology.