King
Vascular Revascularization Device
The device is designed for use in the neurovasculature such as the internal carotid artery, M1 and M2 segments of the middle cerebral artery, basilar, and vertebral arteries.
Rapid and Innovative Technology for Ischemic Stroke Treatment
The KING Vascular Revascularization Device is designed to restore blood flow in patients who have had an ischemic stroke due to large intracranial vascular occlusion.
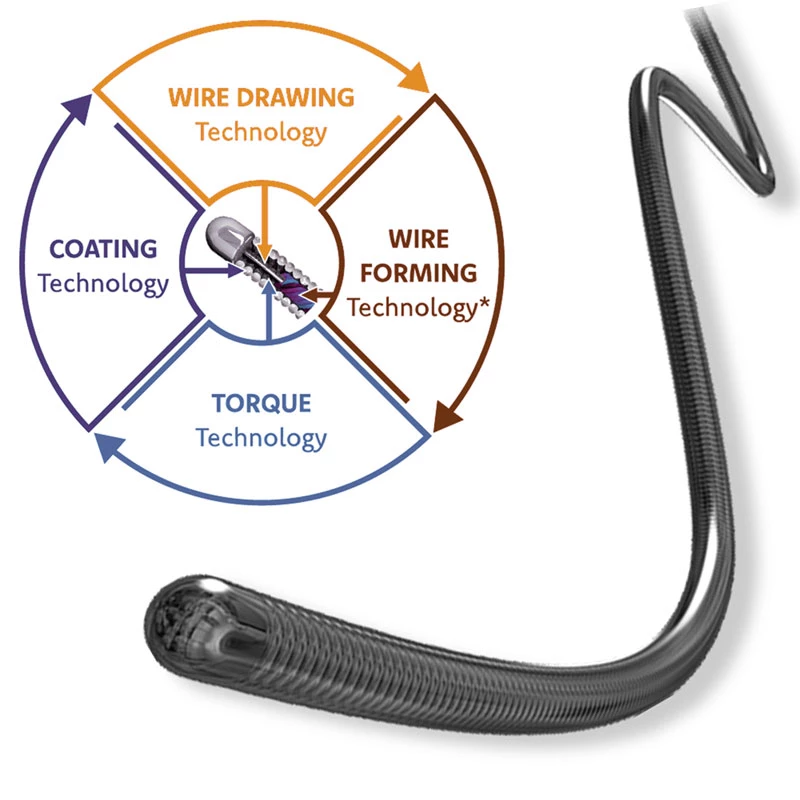
Featuring an overlapping stent-based technological design, the INVAMED Revascularization device provides an innovative technology to improve flow, remove clot and rejuvenate neurological tissue.
The intracranial vascular revascularization device is a self-expanding nitinol device designed to correct blood flow in patients experiencing an acute ischemic stroke due to cranial vessel occlusion. The device is designed to be used in neurovascular structures such as the middle carbide artery, anterior cerebral artery, basilar and vertebral arteries internal carotid artery, and M1 and M2 segments. The revascularization device consists of a caliber design made of nitinol and a push wire attached to the nitinol scaffolding. Supplied preloaded in the introducer sheath. The device is designed to be delivered through a microcatheter placed over the clot and removed with the clot to allow revascularization of the occluded intracranial vessel. The revascularization procedure uses a widely accepted method of administration in which the arterial system is accessed through the femoral artery in the groin. The device is then guided into the brain using standard endovascular techniques.








Detailed specifications
Designed with an optimized delivery system - produces lower delivery force for improved procedural efficiency and smooth navigation through even the complex anatomy.
The treatment approaches using thrombectomy devices in recanalization, and efficient emergency stroke workflow processes have opened new frontiers in managing patients with an acute ischemic stroke.
What are ischemic stroke & carotid artery disease?
An ischemic stroke is a type of stroke caused by a blockage of blood flow within a blood vessel in the brain. The brain cells downstream of the blockage are deprived of oxygen and nutrients and will quickly die if left untreated. The death of a certain amount of brain cells can result in permanent disability or even death.
Carotid artery disease is a narrowing of the carotid arteries. The disease decreases the amount of blood flow to the brain and increases the risk of stroke.
Ischemic Stroke
Ischemic Stroke (IS) occurs as a result of a clot in the artery-blocking the flow of blood to the brain leading to dysfunction or death of the brain tissue.
Carotid Artery Disease
Carotid artery disease is caused by a buildup of plaque in carotid arteries that deliver blood to the brain. Plaque is the deposit of cholesterol, fatty substances, cellular waste products, calcium, and fibrin (a clotting material in the blood) along the wall of a vessel.
DESIGNED TO RESTORE BLOOD FLOW IN PATIENTS WITH ISCHEMIC STROKE
The KingⓇ is consist of an expandable design made of nitinol and a pusher wire attached to the nitinol scaffolding. The KingⓇ is supplied preloaded to the introducer sheath. The device is designed to be delivered via a microcatheter placed on the clot and removed with the clot to ensure revascularization of the occluded intracranial vessel. The revascularization procedure uses a widely accepted model of control where the arterial system is accessed through the femoral artery in the groin. The device is then directed to the brain using standard
KingⓇ4-20 and 6-30 for large thrombus load in carotid “T” and proximal MCA occlusions
- KingⓇ 4-20 for treatment of distal MCA occlusions
- Proven safety and efficacy with excellent long-term outcome
- Unique proximal “ring” design and helical slit provide balanced radial force distribution
Close apposition to the vessel wall during the entire retrieval process
Platinum markers at the proximal and distal ends of the basket are visualized under fluoroscopy.
Fully visible and designed specifically for removing a thrombus in patients experiencing an ischemic stroke.
Rapid and Innovative Technology for Ischemic Stroke Treatment
The INVAMED KingⓇ Vascular Revascularization Device is designed to restore blood flow in patients who have had an ischemic stroke due to large intracranial vascular occlusion.
Featuring an overlapping stent-based technological design, the INVAMED Revascularization device provides an innovative technology to improve flow, remove the clot and rejuvenate neurological tissue.
| Order Number | Description | Diameter | Lenght | Vessel Diameter | Convenient microcatheter Inner Diameter (in) |
| NR1164 | KingⓇVascular Revascularization Device K1 | 4.0 | 180 | 2.0 – 4.0 | 0.021 |
| NR1165 | KingⓇVascular Revascularization Device K2 | 4.0 | 180 | 2.0 – 4.0 | 0.021 |
| NR1166 | KingⓇ Vascular Revascularization Device B1 | 6.0 | 180 | 3.0 – 5.5 | 0.027 |
| NR1167 | KingⓇVascular Revascularization Device B2 | 6.0 | 180 | 3.0 – 5.5 | 0.027 |